Alzheimer’s Disease: Recent Research & Therapeutic Advances
Scientific article by Nina Culum, MSc
Alzheimer’s disease is a slow progressive neurodegenerative disease affecting an estimated 6.2 million Americans aged 65 and older in 2021 alone [1]. Although its prevalence increases with age, researchers stress that Alzheimer’s disease is not a normal part of aging. As the number of Americans aged 65 and older is projected to grow rapidly over the next few decades, Alzheimer’s disease prevalence is expected to increase as well. The projected increase in individuals living with Alzheimer’s disease is of great concern to researchers, particularly given the lack of a single diagnostic test during early disease stages and the fact that there is currently no cure.
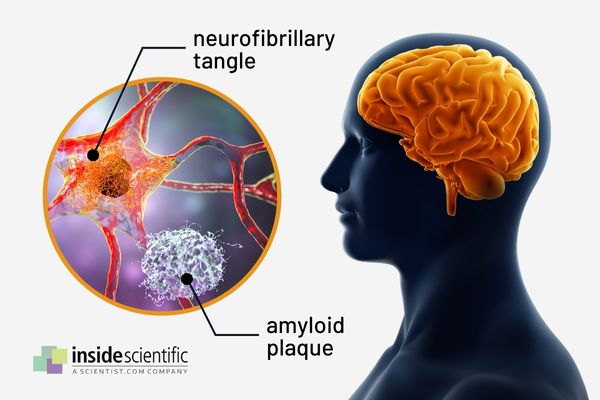
The Alzheimer’s brain is characterized by the presence of extraneuronal amyloid plaques and intraneuronal neurofibrillary tangles, which are both abnormal structures containing highly insoluble and densely packed filaments (Figure 1) [2]. Amyloid plaques are formed by the accumulation of beta-amyloid (Aβ) peptides, most notably containing 40 (Aβ40) and 42 (Aβ42) amino acids, while neurofibrillary tangles are composed of hyperphosphorylated tau proteins [3]. The accumulation of these plaques and tangles is accompanied by neuronal damage and coincides with symptom onset. Early symptoms of Alzheimer’s disease can include apathy and depression, while impaired communication, confusion, poor judgment, and behavioral changes are commonly reported at later stages of disease [1].
While Aβ is the most prominent target in pharmacological clinical trials, additional important features of Alzheimer’s disease include neuroinflammation, mitochondrial dysfunction, endoplasmic reticulum stress, and abnormal calcium influx [4]. A major genetic risk factor for Alzheimer’s disease is the ε4 allele of the apolipoprotein E gene (APOE ε4). Individuals with APOE ε4 are more likely to have Aβ accumulation and Alzheimer’s disease at a younger age than those with APOE ε2 or ε3 [5].
Biomarkers of Alzheimer’s Disease Pathology
No single diagnostic test currently exists for Alzheimer’s disease. Standard Alzheimer’s disease testing is based on a battery of tools and assessments, including structural imaging to rule out other conditions that may cause similar symptoms but require different treatments. The most widely used biomarker in clinical studies for Alzheimer’s disease diagnosis is Aβ measured in cerebrospinal fluid (CSF) and by positron emission tomography (PET) [3].
Alzheimer’s disease CSF is characterized by an approximate 50% reduction in the concentration of Aβ42 [6], and increased diagnostic accuracy can be achieved by measuring CSF Aβ42/Aβ40 ratios. This ratio is nearly completely concordant with amyloid PET imaging, which has undergone extensive standardization [7]. However, CSF sampling is an invasive procedure, and amyloid PET is expensive, has limited availability, and is associated with a non-negligible radiation exposure [3]. Therefore, the identification and validation of blood biomarkers for Aβ pathology would greatly improve diagnostics for patients with cognitive impairment. Plasma Aβ42/Aβ40 ratios have already been shown to reflect cerebral Aβ pathology with high accuracy compared to amyloid PET and CSF Aβ42/Aβ40 ratios [8].
PrecivityAD is the first approved blood test designed to aid in Alzheimer’s disease detection decades before symptom onset. This method quantifies plasma Aβ and APOE levels by liquid chromatography with tandem mass spectrometry [9]. The results, which also account for a patient’s age, provide a probability score of a patient having an amyloid-positive brain scan.
CSF tau may also be regarded as a predictive marker of Alzheimer’s disease-type neurodegeneration [3]. Standardization work for tau imaging is still ongoing, but it could become a valuable tool for evaluating the efficacy of amyloid, tau, or combination therapy. Furthermore, tau PET has demonstrated the ability to detect the pharmacodynamic effects of disease-modifying drugs targeting both Aβ and tau pathology [10].
In this webinar, Dr. Jagirdar presented how acute activation of the thalamic reticular nucleus improves sleep quality in a mouse model of Alzheimer’s disease. He also discussed how chronic TRN activation improved sleep quality for the duration of treatment and decreased β-amyloid plaque accumulation in mice. WATCH NOW
Dr. Fiona Harrison discusses how dietary deficiency and exposure to toxins can impact glutamate uptake and clearance, and the subsequent changes in neural signaling can be detected through altered EEG activity and performance on learning and memory tasks. WATCH NOW
Limitations of Conventional Alzheimer’s Disease Therapies
Since there is currently no cure for Alzheimer’s disease, a vast majority of pharmacologic therapies that are approved by the Food and Drug Administration (FDA) for Alzheimer’s disease are based on symptom management using cholinesterase inhibitors, N-methyl-D-aspartate antagonists, and combination therapy [4]. These medications only temporarily improve cognitive functions, and the effectiveness of these drugs varies among individuals. The slow development of effective Alzheimer’s disease treatments has been influenced by many factors, including gaps in knowledge of the precise molecular processes that cause Alzheimer’s disease and the prevalence of mixed dementia that can complicate treatment decisions [1]. Furthermore, current therapies may begin too late for the reversal of Alzheimer’s disease pathology, as clinical symptoms typically develop almost 20 years after the first appearance of pathological signs.
Emerging Therapies Targeting Alzheimer’s Disease Pathology
In 2021, the FDA granted accelerated approval for aducanumab, the first new medication for Alzheimer’s disease since 2003 and the first medication targeting Aβ plaques in the brain [11]. Currently, aducanumab has only been tested in individuals with mild cognitive impairment (MCI) or early Alzheimer’s disease. The decision to approve aducanumab was highly controversial in the scientific community, with researchers citing inconclusive phase III clinical results and a lack of evidence that the benefits outweigh the risk of the treatment [12, 13]. FDA approval was based on the observation that plaque reduction is reasonably likely to result in clinical benefit as well as the urgent need for development in this area of medicine [11]. The stipulation of the accelerated approval pathway is that a phase IV post-marketing randomized controlled trial is required by 2030 to verify the clinical benefits.
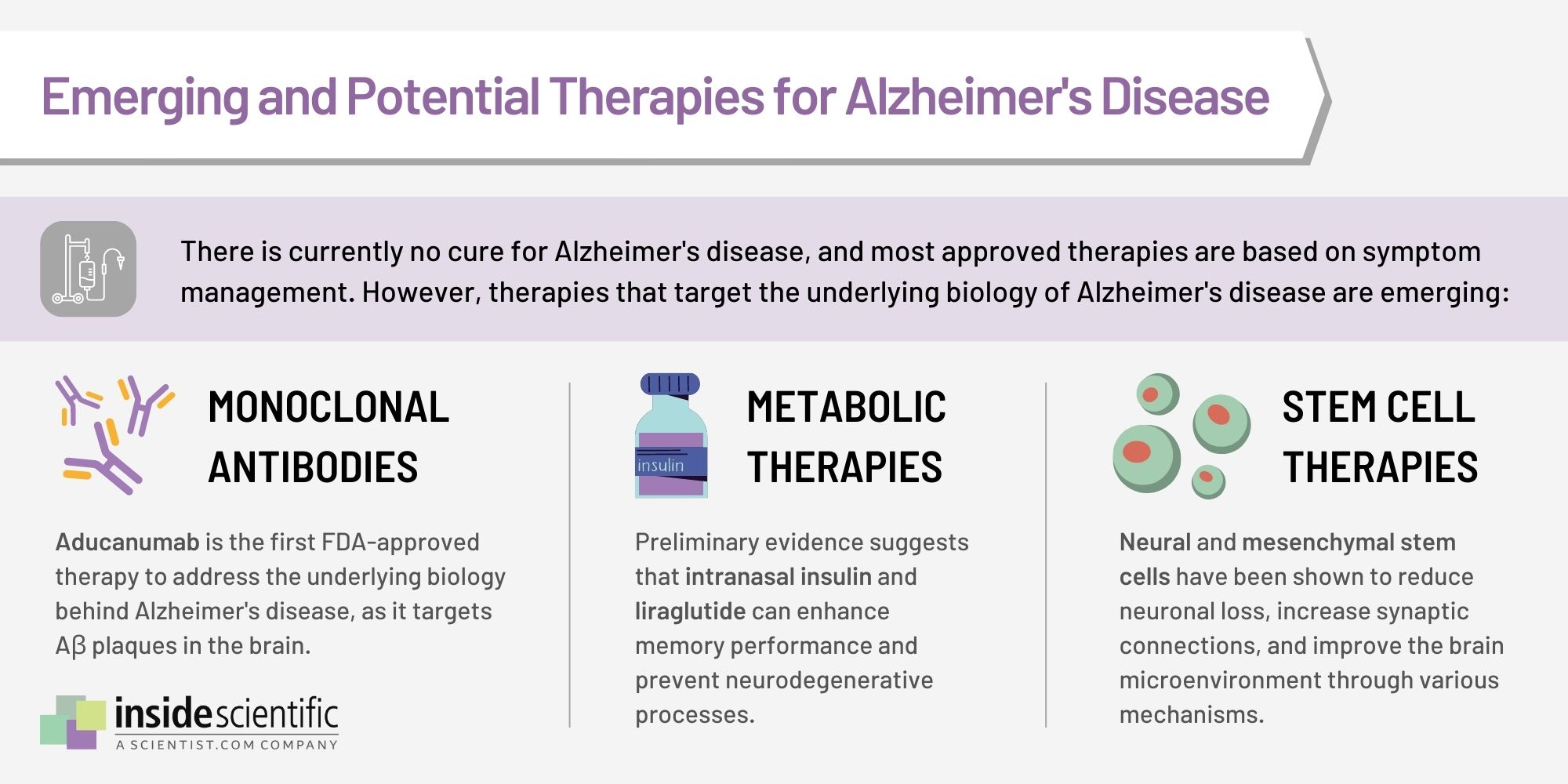
Since the approval of aducanumab, other anti-Aβ monoclonal antibodies such as gantenerumab, lecanemab, and donanemab have been granted breakthrough therapy designation by the FDA [14]. The breakthrough therapy designation program is intended to accelerate the development and review of medications for serious or life-threatening conditions. Researchers note that competing anti-Aβ therapies can drive down medication prices and increase the urgency of clinical trials to prove drug efficacy [14]. Alternative Alzheimer’s disease therapies being explored at preclinical and early clinical stages include metabolic and stem cell therapies (Figure 2).
Metabolic therapies for Alzheimer’s disease treatment
Brain insulin signaling contributes to memory function, and several studies have indicated that intranasal insulin (INI) enhances memory performance in individuals with Alzheimer’s disease [15]. In a mouse model of Alzheimer’s disease, INI improved insulin signaling activation by ameliorating biliverdin reductase-A activation, and was associated with reduced tau phosphorylation and Aβ in the brain as well as improved cognitive function [16]. More recently, it has been suggested that the neuroprotective effects of INI on cognition could be due to microRNA regulation in the brain, as shown in a rat model of Alzheimer’s disease [17]. In addition to these mechanistic studies, clinical studies have reported that INI treatment reduces white matter hyperintensity volume progression and modulates CSF markers of neuroinflammation in patients with MCI and Alzheimer’s disease [18, 19].
Liraglutide, among other anti-diabetes medications, has been widely assessed in animal models of Alzheimer’s disease [20, 21]. Early research demonstrated that liraglutide could prevent neurodegenerative processes in mouse models by reducing tau phosphorylation, synaptic plasticity, Aβ plaque formation, and inflammation [22, 23]. More recently, liraglutide has been reported to exert neuroprotective effects in an Alzheimer’s mouse model, and could serve as an anti-inflammatory and anti-amyloid prophylactic therapy for Alzheimer’s disease [24]. Liraglutide treatment has also been shown to improve cognitive function in a rat model of diabetes [25].
Dr. Marco Weiergräber and Dr. Jennifer Teske present telemetry implant procedure for wireless recording of EEG in rodents and share novel case studies focused on studying neural recordings to investigate sleep, energy expenditure and Alzheimer’s disease. WATCH NOW
Stem cell therapies for Alzheimer’s disease treatment
Some researchers note that stem cell therapies including neural stem cells (NSCs) and mesenchymal stem cells (MSCs) may be more promising and beneficial Alzheimer’s disease treatments than traditional therapies since they can reduce neuronal loss, increase synaptic connections, and fundamentally improve the brain microenvironment [26, 27]. Specific stem cell mechanisms of action in treating Alzheimer’s disease include anti-inflammatory responses, replacing injured or lost neuronal cells, secreting neurotrophic factors, producing anti-amyloid proteins, promoting the activation of endogenous stem cells, and improving neuronal metabolic activity in the brain [27].
One recent study reported that intranasal transplantation of human NSCs (hNSCs) into the brains of mice could attenuate Aβ accumulation, ameliorate neuroinflammation and synaptic loss, and improve cognitive deficits [28]. Extracellular vesicles (EVs) derived from NSCs that continuously express neprilysin have been shown to improve Aβ clearance and neural regeneration in a murine Alzheimer’s disease model [29]. Additionally, hNSC-derived EVs have been recently reported to restore fear extinction memory consolidation, protect against synaptic loss, improve cognition, and reduce anxiety-related behaviors and Aβ plaque accumulation in a mouse model of Alzheimer’s disease [30].
Recently, a single intravenous MSC dose in an Alzheimer’s mouse model was reported to attenuate neuroinflammation, while multiple MSC doses decreased pathological tau phosphorylation [31]. MSC-derived EVs have also been shown to slow down Alzheimer’s disease pathogenesis in mice, potentially by modulating inflammation during disease development [32]. Furthermore, it has been demonstrated that MSC-derived exosomes can significantly improve brain glucose metabolism and cognitive function in a mouse model of Alzheimer’s disease [33].
How is Alzheimer’s disease studied in animals?
Many symptoms of Alzheimer’s disease are caused by pathology in organs outside of the central nervous system, so additional non-neurological outcome measures are valuable in Alzheimer’s research [34]. For example, many researchers have employed telemetry for wireless electroencephalogram (EEG) recordings in animal models of Alzheimer’s disease. Since EEG is directly translatable to humans, it may also serve as an early Alzheimer’s diagnostic biomarker [35]. Recently, Wilcox et al. employed the PhysioTel HD-X02 telemetry device by Data Sciences International (DSI) alongside Ponemah software to monitor EEG in Alzheimer’s disease mice, and determined that the cognitive decline observed in Alzheimer’s disease patients could be due to hyperexcitability and seizure activity rather than other pathologies [35]. Przybyla et al. have also employed DSI telemetry and NeuroScore software to monitor EEG activity and characterize the effects of transgenic human tau expression on neuronal network function in mice [36]. In this study, memory deficits were found to be associated with progressive neuronal network dysfunction in the hippocampus [36].
Sleep is another valuable outcome measure in Alzheimer’s research, as changes in sleep/wake activity may be risk factors for Alzheimer’s disease [37]. Gould et al. recently determined that M1-positive allosteric modulators may be beneficial in normalizing sleep deficits with minimal adverse effects using DSI telemetry devices for small and large animals, and further demonstrated the utility of EEG as a translational Alzheimer’s marker [37]. Cognitive disturbances, sleep cycles, and epileptiform activity have also been wirelessly monitored via DSI implantable telemetry by Zegarra-Valdivia et al. in a mechanistic study of how insulin-like growth factor I affects Alzheimer’s disease pathology in mice [38]. Additionally, Alzheimer’s disease patients often exhibit respiratory control dysfunction, and disordered breathing can result in periods of hypoxia during sleep [39]. Since these underlying mechanisms are unknown, Brown et al. defined the pathophysiological respiratory response of Alzheimer’s rats to two hours of acute hypoxia achieved with a whole-body, flow-through plethysmography chamber by DSI, and further analyzed hypoxia-induced neuronal activation in areas of the brainstem [39].
Monitoring animal behavior by video tracking has also been shown to be beneficial in preclinical Alzheimer’s research. Hu et al. examined the effects of a small molecule integrated stress response inhibitor (ISRIB) on the facilitation of long-term depression and behavioral deficits in rats, wherein animal behavior was monitored using Panlab’s SMART video tracking system [40]. Data from these experiments demonstrated that ISRIB exhibits promising protective effects on Aβ-induced spatial learning and memory deficits, and may protect against synaptic and cognitive disruption in early Alzheimer’s disease [40].
For more information on in vivo research approaches for studying neurological disease and disorders, download this white paper from DSI.
Harvard Bioscience solutions for preclinical Alzheimer’s research
Harvard Bioscience empowers Alzheimer’s research with a complete package of laboratory tools, including systems for identifying biomarkers and studying channelopathies, synaptic neurotransmission, neural network connectivity, dynamics, and synchronization:
- BTX’s electroporation systems simplify electroporation-enhanced in vivo gene transfer as well as the generation of genetically manipulated models
- Biochrom’s spectrophotometers and microplate readers support the evaluation of molecular biomarkers with a wide array of UV-visible and colorimetric assays for nucleic acid and protein quantification, as well as cell viability and proliferation assays
- Warner’s imaging chambers facilitate live-cell imaging investigations in cell cultures and brain slices
- CMA’s reliable microdialysis probes help elucidate brain neurophysiology as well as released neurotransmitters, metabolites, and other endogenous compounds including Aβ peptides and tau proteins
- Wireless monitoring of brain activity in freely moving animals can be performed using Multi-Channel Systems’ headstages for high-resolution EEG recordings or DSI’s telemetry implants for longitudinal EEG examinations
- Panlab’s wide array of behavioral research products including operant conditioning chambers and video tracking solutions enable the analysis of cognition, learning, and memory processes
Perspectives, Outlooks, and Additional Resources
While certain risk factors for Alzheimer’s disease such as age, genetics, and family history of disease cannot be controlled, researchers are investigating modifiable risk factors to slow or reverse disease development. For example, individuals with higher physical activity are less at risk of developing Alzheimer’s disease [41], while individuals who experience sleep disturbance are more at risk of Aβ accumulation [42]. Diet also appears to be extremely important in Alzheimer’s prevention. The Mediterranean diet in particular is associated with a reduced susceptibility to Alzheimer’s disease and lower Aβ levels [43, 44].
The development of Alzheimer’s disease therapies, especially those targeting the reversal of Alzheimer’s disease pathology, will have a tremendous impact on the quality of life of individuals with Alzheimer’s disease and their caregivers. Furthermore, effective therapies could lower the financial burden associated with acute and long-term care for individuals with Alzheimer’s disease. To learn more about recent developments in Alzheimer’s research, explore our latest webinars on the vascular contributions to late-onset Alzheimer’s disease and dementia, and the electrophysiology of human native receptors in neurological and mental disorders including Alzheimer’s disease.
About the Author
About the Author

Nina Culum graduated from the University of Western Ontario with a Master of Science in physical and analytical chemistry. During her graduate studies, she fabricated plasmonic nanohole arrays to capture extracellular vesicles and detect cancer by surface-enhanced Raman spectroscopy. Prior to attending UWO, Nina completed her Bachelor of Science in chemistry at the University of Waterloo.
References
- 2021 Alzheimer’s disease facts and figures. Alzheimers Dement. 2021;71(3):327-406. DOI: 10.1002/alz.12328.
- Kumar A, Nemeroff CB, Cooper JJ, Widge A, Rodriguez C, Carpenter L, et al. Amyloid and tau in Alzheimer’s disease: biomarkers or molecular targets for therapy? Are we shooting the messenger? Am J Psychiatry. 2021;178(11):1014-25. DOI: 10.1176/appi.ajp.2021.19080873.
- Zetterberg H, Bendlin BB. Biomarkers for Alzheimer’s disease—preparing for a new era of disease-modifying therapies. Mol Psychiatry. 2021;26(1):296-308. DOI: 10.1038/s41380-020-0721-9.
- Domingues R. Pereira C, Cruz MT, Silva A. Therapies for Alzheimer’s disease: a metabolic perspective. Mol Genet Metab. 2021;132(3):162-72. DOI: 10.1016/j.ymgme.2021.01.011.
- Jansen WJ, Ossenkoppele R, Knol DL, Tijms BM, Scheltens P, Verhey FRJ, et al. Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA. 2015;313(19):1924-38. DOI: 10.1001/jama.2015.4668.
- Olsson B, Lautner R, Andreasson U, Ohrfelt A, Portelius E, Bjerke M, et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: a systematic review and meta-analysis. Lancet Neurol. 2016;15(7):673-84. DOI: 10.1016/S1474-4422(16)00070-3.
- Hansson O, Lehmann S, Otto M, Zetterberg H, Lewczuk P. Advantages and disadvantages of the use of the CSF Amyloid β (Aβ) 42/40 ratio in the diagnosis of Alzheimer’s disease. Alzheimers Res Ther. 2019;11(1):34. DOI: 10.1186/s13195-019-0485-0.
- Schindler SE, Bollinger JG, Ovod V, Mawuenyega KG, Li Y, Gordon BA, et al. High-precision plasma β-amyloid 42/40 predicts current and future brain amyloidosis. Neurology. 2019;93(17):e1647-59. DOI: 10.1212/WNL.0000000000008081.
- Kirmess KM, Meyer MR, Holubasch MS, Knapik SS, Hu Y, Jackson EN, et al. The PrecivityAD™ test: accurate and reliable LC-MS/MS assays for quantifying plasma amyloid beta 40 and 42 and apolipoprotein E proteotype for the assessment of brain amyloidosis. Clin Chim Acta. 2021;519:267-75. DOI: 10.1016/j.cca.2021.05.011.
- Schöll M, Maass A, Mattsson N, Ashton NJ, Blennow K, Zetterberg H, et al. Biomarkers for tau pathology. Mol Cell Neurosci. 2019;97:18-33. DOI: 10.1016/j.mcn.2018.12.001.
- U.S. Food & Drug Administration [Internet]. FDA grants accelerated approval for Alzheimer’s drug; 2021 Jun 7 [cited 2021 Nov 19]. Available from: https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-alzheimers-drug.
- Mullard A. Controversial Alzheimer’s drug approval could affect other diseases. Nature. 2021;595(7866):162-63. DOI: 10.1038/d41586-021-01763-9.
- Perlmutter JS. Aducanumab: look before leaping. Nat Med. 2021;27(9):1499. DOI: 10.1038/s41591-021-01477-5.
- Lalli G, Schott JM, Hardy J, de Strooper B. Aducanumab: a new phase in therapeutic development for Alzheimer’s disease? EMBO Mol Med. 2021;13(8):e14781. DOI: 10.15252/emmm.202114781.
- Hallschmid, M. Intranasal insulin for Alzheimer’s disease. CNS Drugs. 2021;35:21-37. DOI: 10.1007/s40263-020-00781.
- Barone E, Tramutola A, Tiriani F, Calcagnini S, Di Domenico F, Ripoli C, et al. Biliverdin reductase-A mediates the beneficial effects of intranasal insulin in Alzheimer disease. Mol Neurobiol. 2019;56(4):2922-43. DOI: 10.1007/s12035-018-1231-5.
- Bazrgar M, Khodabakhsh P, Dargahi L, Mohagheghi F, Ahmadiani A. MicroRNA modulation is a potential molecular mechanism for neuroprotective effects of intranasal insulin administration in amyloid βeta oligomer induced Alzheimer’s like rat model. Exp Gerontol. 2022;164:111812. DOI: 10.1016/j.exger.2022.111812.
- Kellar D, Lockhart SN, Aisen P, Raman R, Rissman RA, et al. Intranasal insulin reduces white matter hyperintensity progression in association with improvements in cognition and CSF biomarker profiles in mild cognitive impairment and Alzheimer’s disease. J Prev Alzheimer’s Dis. 2021;8:240-8. DOI: 10.14283/jpad.2021.14.
- Kellar D, Register T, Lockhart SN, Aisen P, Raman R, et al. Intranasal insulin modulates cerebrospinal fluid markers of neuroinflammation in mild cognitive impairment and Alzheimer’s disease: a randomized trial. Sci Rep. 2022;12:1346. DOI: 10.1038/s41598-022-05165-3.
- Michailidis M, Tata DA, Moraitou D, Kavvadas D, Karachrysafi S, et al. Antidiabetic drugs in the treatment of Alzheimer’s disease. Int J Mol Sci. 2022;23(9):4641. DOI: 10.3390/ijms23094641.
- Vargas-Soria M, Carranza-Naval MJ, del Marco A, Garcia-Alloza M. Role of liraglutide in Alzheimer’s disease pathology. Alz Res Therapy. 2021;13:112. DOI: 10.1186/s13195-021-00853-0.
- McClean PL, Parthsarathy V, Faivre E, Hölscher C. The diabetes drug liraglutide prevents degenerative processes in a mouse model of Alzheimer’s disease. J Neurosci. 2011;31(17):6587-94. DOI: 10.1523/JNEUROSCI.0529-11.2011.
- Ma D-L, Chen F-Q, Xu W-J, Yue W-Z, Yuan G, Yan Y. Early intervention with glucagon-like peptide 1 analog liraglutide prevents tau hyperphosphorylation in diabetic db/db mice. J Neurochem. 2015;135(2):301-8. DOI: 10.1111/jnc.13248.
- Paladugu L, Gharaibeh A, Kolli N, Learman C, Hall TC, et al. Liraglutide has anti-inflammatory and anti-amyloid properties in streptozotocin-induced and 5xFAD mouse models of Alzheimer’s disease. Int J Mol Sci. 2021;22(2):860. DOI: 10.3390/ijms22020860.
- Sedky AA. Improvement of cognitive function, glucose and lipid homeostasis and serum osteocalcin levels by liraglutide in diabetic rats. Fundam Clin Pharmacol. 2021;35:989-1003. DOI: 10.1111/fcp.12664.
- Salwa LK. Engrafted stem cell therapy for Alzheimer’s disease: a promising treatment strategy with clinical outcome. J Control Release. 2021;338:837-57. DOI: 10.1016/j.jconrel.2021.09.007.
- Si Z, Wang X. Stem cell therapies in Alzheimer’s disease: applications for disease modeling. J Pharmacol Exp Ther. 2021;377(2):207-17. DOI: 10.1124/jpet.120.000324.
- Lu M-H, Ji W-L, Chen H, Sun Y-Y, Zhao X-Y, et al. Intranasal transplantation of human neural stem cells ameliorates Alzheimer’s disease-like pathology in a mouse mode. Front Aging Neurosci. 2021;13:650103. DOI: 10.3389/fnagi.2021.650103.
- Huang D, Cao Y, Yang X, Liu Y, Zhang Y, et al. A nanoformulation-mediation multifunctional stem cell therapy with improved beta-amyloid clearance and neural regeneration for Alzheimer’s disease. Adv Mater. 2021;33:2006357. DOI: 10.1002/adma.202006357.
- Apodaca LA, Baddour AAD, Garcia Jr C, Alikhani L, Giedzinski E, et al. Human neural stem cell-derived extracellular vesicles mitigate hallmarks of Alzheimer’s disease. Alz Res Therapy. 2021;13:57. DOI: 10.1186/s13195-021-00791-x.
- Neves AF, Camargo C, Premer C, Hare JM, Baumel BS, et al. Intravenous administration of mesenchymal stem cells reduces Tau phosphorylation and inflammation in the 3xTg-AD mouse model of Alzheimer’s disease. Exp Neurol. 2021;341:113706. DOI: 10.1016/j.expneurol.2021.113706.
- Cone AS, Yuan X, Sun L, Duke LC, Vreones MP, et al. Mesenchymal stem cell-derived extracellular vesicles ameliorate Alzheimer’s disease-like phenotypes in a preclinical mouse model. Theranostics. 2021;11(17):8129-42. DOI: 10.7150/thno.62069.
- Chen Y-A, Lu C-H, Ke C-C, Chiu S-J, Jeng F-S, et al. Mesenchymal stem cell-derived exosomes ameliorate Alzhimer’s disease pathology and improve cognitive deficits. Biomedicines. 2021;9(6):594. DOI: 10.3390/biomedicines9060594.
- Data Sciences International. Alzheimer’s Research. [cited 2022 May 31]. Available from: https://www.datasci.com/solutions/neuroscience/alzheimer’s-disease
- Wilcox JM, Consoli DC, Tienda AA, Dixit S, Buchanan RA, et al. Altered synaptic glutamate homeostasis contributes to cognitive decline in young APP/PSEN1 mice. Neurobiol Dis. 2021;158:105486. DOI: 10.1016/j.nbd.2021.105486.
- Przybyla M, van Eersel J, van Hummel A, van der Hoven J, Sabale M, et al. Onset of hippocampal network aberration and memory deficits in P301S tau mice are associated with an early gene signature. Brain. 2020;143(6):1889-904. DOI: 10.1093/brain/awaa133.
- Gould RW, Russell JK, Nedelcovych MT, Bubser M, Blobaum AL, et al. Modulation of arousal and sleep/wake architecture by M1 PAM VU0453595 across young and aged rodents and nonhuman primates. Neuropsychopharmacology. 2020;45:2219-28. DOI: 10.1038/s41386-020-00812-7.
- Zegarra-Valdivia JA, Santi A, Fernández de Sevilla ME, Nuñez A, Torres Aleman I. Serum insulin-like growth factor I deficiency associates to Alzehimer’s disease co-morbidities. J Alzheimer’s Dis. 2019;69(4):979-87. DOI: 10.3233/JAD-190241.
- Brown AG, Thapa M, Hooker IV JW, Ostrowski TD. Impaired chemoreflex correlates with decreased c-Fos in respiratory brainstem centers of the streptozotocin-induced Alzheimer’s disease rat model. Exp Neurol. 2019;311:285-92. DOI: 10.1016/j.expneurol.2018.10.012.
- Hu Z, Yu P, Zhang Y, Yang Y, Zhu M, et al. Inhibition of the ISR abrogates mGluR5-dependent long-term depression and spatial memory deficits in a rat model of Alzheimer’s disease. Transl Psychiatry. 2022;12:96. DOI: 10.1038/s41398-022-01862-9.
- Meng Q, Lin M-S, Tzeng I-S. Relationship between exercise and Alzheimer’s disease: a narrative literature review. Front Neurosci. 2020;14:131. DOI: 10.3389/fnins.2020.00131.
- Winer JR, Mader BA, Kumar S, Reed M, Baker SL, Jagust WJ, et al. Sleep disturbance forecasts β-amyloid accumulation across subsequent years. Curr Biol. 2020;30(21):4291-98. DOI: 10.1016/j.cub.2020.08.017.
- Morris MC, Tangney CC, Wang Y, Sacks FM, Bennett DA, Aggarwal NT. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 2015;11(9):1007-14. DOI: 10.1016/j.jalz.2014.11.009.
- Berti V, Walters M, Sterling J, Quinn CG, Logue M, Andrews R, et al. Mediterranean diet and 3-year Alzheimer brain biomarker changes in middle-aged adults. Neurology. 2018;90(20):e1789-98. DOI: 10.1212/WNL.0000000000005527.