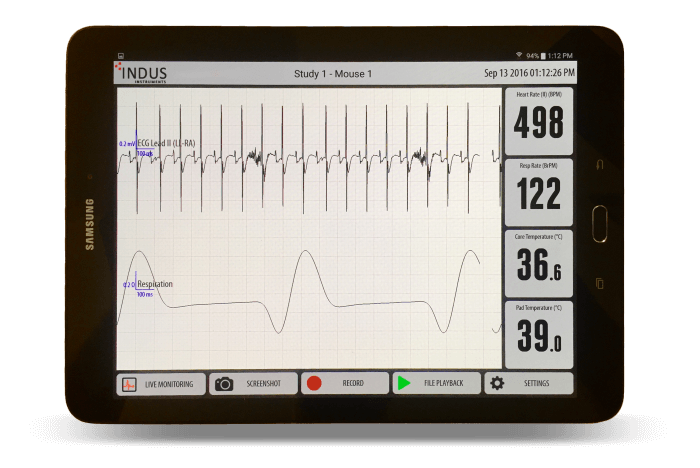
On January 15, 2014, InsideScientific and Indus Instruments hosted a webinar about the importance of multimetric surgical monitoring to ensure survival rate and more consistent results. Speaker Graham Sattler of Indus Instruments discussed essentials of surgical monitoring and described the signal integration of the Mouse Monitor S. Next, Dr. Corey Reynolds outlined NIH guidelines and requirements, highlighted the importance of integrated surgical monitoring systems, and describes applications in the Mouse Phenotyping Core. Dr. Merry Lindsey then discussed applications in the lab, using as an example her research in cardiac remodelling following myocardial infarction.
Follow the links below to access key educational points of the webinar…
Dr. Merry Lindsey
- 26:08 Myocardial infarction (MI) and post-MI cardiac remodelling: current research areas
- 31:35 Example mouse recording for a pre- and post-MI setting
- 33:20 Critical needs and best practices for in vivo models and surgery
Q&A
- 37:16 Are there other research models this could be used for?
- 38:28 Can the Mouse Monitor S be integrated with other PV loop systems?
- 40:28 In the event someone has a lab with multiple platforms, how would you suggest arranging that?
- 43:02 Can you describe the process and the options available through the Mouse Monitor directly for ECG filtering?
- 46:00 Could a signal be left ‘untouched’ so it could be passed on to an external data acquisition system so data management could be handled downstream?
- 47:08 Could you talk a bit about the pulse oximetry module? What system features have been built in to ensure optimal signal clarity, because there are so many signals pulled to one device?
SPEAKERS:
- Graham Sattler
Product Development Manager, Indus Instruments
LinkedIn Profile
ResearchGate Profile - Corey Reynolds, PhD
Assistant Professor, Advanced Technology Cores, Baylor College of Medicine
Baylor College of Medicine profile
LinkedIn Profile
ResearchGate Profile - Merry L. Lindsey, PhD
Professor of Physiology and Medicine, Director Mississippi Center for Heart Research
University of Mississippi Medical Center profile
LinkedIn
ResearchGate
Google Scholar author search
ASSOCIATED RESOURCES:
REFERENCES & CITATIONS:
- Guide for the care and use of laboratory animals. (2011). Washington, D.C.: National Academies Press.
- Thygesen, K., Alpert, J. S., & White, H. D. (2007). Universal Definition of Myocardial Infarction. Journal of the American College of Cardiology, 50(22), 2173-2195. doi:10.1016/j.jacc.2007.09.011
- Yabluchanskiy, A., Chilton, R. J., & Lindsey, M. L. (2013). Left ventricular remodeling: one small step for the extracellular matrix will translate to a giant leap for the myocardium. Congestive Heart Failure (Greenwich, Conn.), 19(4), E5–E8. http://doi.org/10.1111/chf.12023
- Lindsey, M. L., Escobar, G. P., Dobrucki, L. W., Goshorn, D. K., Bouges, S., Mingoia, J. T., . . . Spinale, F. G. (2006). Matrix metalloproteinase-9 gene deletion facilitates angiogenesis after myocardial infarction. American Journal of Physiology-Heart and Circulatory Physiology, 290(1). doi:10.1152/ajpheart.00457.2005
- Chiao, Y. A., Zamilpa, R., Lopez, E. F., Dai, Q., Escobar, G. P., Hakala, K., … Lindsey, M. L. (2010). In vivo Matrix Metalloproteinase-7 Substrates Identified in the Left Ventricle Post-Myocardial Infarction Using Proteomics. Journal of Proteome Research, 9(5), 2649–2657. http://doi.org/10.1021/pr100147r
- Ma, Y., Halade, G. V., Zhang, J., Ramirez, T. A., Levin, D., Voorhees, A., … Lindsey, M. L. (2013). Matrix Metalloproteinase-28 Deletion Exacerbates Cardiac Dysfunction and Rupture Following Myocardial Infarction in Mice by Inhibiting M2 Macrophage Activation. Circulation Research, 112(4), 675–688. http://doi.org/10.1161/CIRCRESAHA.111.300502