Dr. Malmstrøm demonstrates how correlations between NMR data and bioactivity can be used to confirm the structure and activity of proteins.
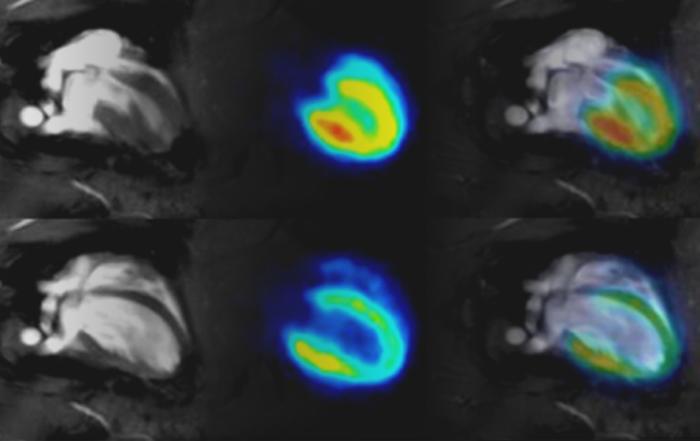
An important aspect in analytical assessment of pharmaceutical proteins is the comprehensive characterization of their higher order structure (HOS). The most sensitive and detailed method for measuring HOS in proteins is unquestionably 2D NMR spectroscopy. It is well-established that the biological functions of proteins depend on their specific structures. However, the quality of bioactive proteins is traditionally assessed indirectly by cell-based potency assays. These assays usually take several days, are laborious, and have considerable variability. In addition, development of suitable assays for oral formulations is facing severe challenges due to excipient interference. The need for a rapid, precise, sensitive, and robust HOS assay has increased since the regulatory agencies are now approving “biosimilar” pharmaceutical proteins based on HOS similarities.
In this fifth and final event in the Bruker COVID-19 Webinar Series, Dr. Joan Malmstrom will demonstrate that NMR can be used to ensure that the structure, and thereby the activity, of a protein is intact. She will do this by focusing on her work that shows how to measure a correlation between bioactivity and NMR data. She will share specific examples of how this can be done by measuring the bioactivity and NMR data on intact samples as well as samples that have been stressed under extreme conditions.
Presenters
CMC Analytical Support
R&D
Novo Nordisk