Scientists present methodology and research findings from neurophysiological studies in head-fixed, behaving mice.
euroscience research in freely behaving animals is inherently challenging for techniques that require a high degree of precision, such as 2-photon imaging, single-cell optogenetics and patch clamp recording. Scientists are forced to use general anesthesia to immobilize animals for single-cell recording and microscopy. Unfortunately, anesthetics have a profound physiological impact that compromises data quality and translational relevance.
In this webinar sponsored by Neurotar, experts present their research utilizing the Mobile HomeCage®, an experimental tool which ensures the stability required for high-precision neurophysiological techniques while allowing mice to navigate and explore their environment.
Case Study #1:
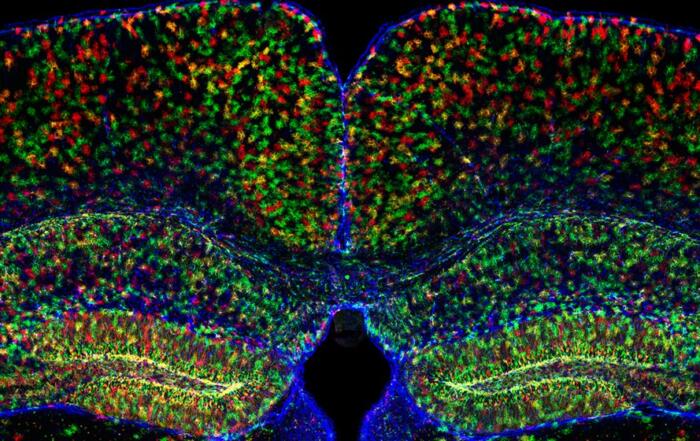
Dr. Sarah Stuart and Dr. Jon Palacios-Filardo of the University of Bristol present their studies combining analysis of goal-directed behavior with whole-cell recordings from the hippocampus of awake mice. The researchers share useful tips for the surgery protocol and for adjusting the head fixation angle in order to facilitate mouse motility and exploratory behavior.
Case Study #2:
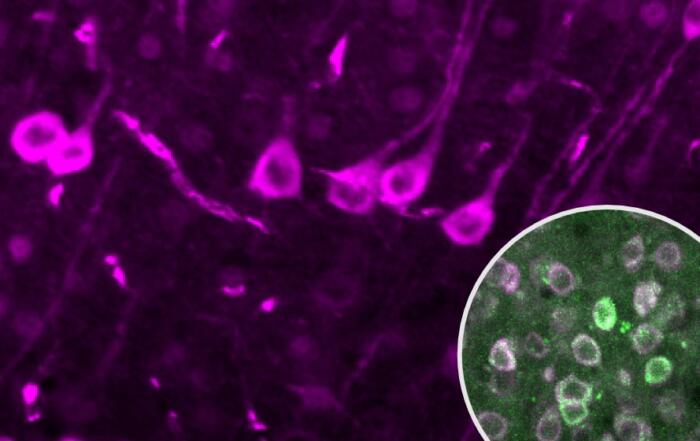
Dr. Alexander Dityatev and Weilun Sun from the German Center for Neurodegenerative Diseases (DZNE) discuss 2-photon imaging of fluorescently labeled microglia in vivo in the context of neurodegenerative disease. They also present their recent data on the effects of different anesthetics on the microglial response to localized laser injury.
Case Study #3:
Dr. Norbert Hájos from the Hungarian Academy of Sciences presents his lab’s research into the amygdala’s role in reward-driven behavior. He shares the challenges of making single-unit recordings using silicon probes during mouse locomotion and subsequent morphological identification of active neurons in the amygdala.
Key Topics Include:
- Requirements for stable single-cell recordings and 2-photon imaging in behaving mice
- Challenges of combining high-precision techniques with behavioral research
- Methodological considerations for improving exploratory behavior in head-fixed mice
- Quantitative analysis of microglial function using 2-photon microscopy in awake mice
- Recording neuronal activity in the amygdala of awake mice followed by morphological identification of recorded neurons
Click to watch the webinar recording. To view the presentation full screen simply click the square icon located in the bottom-right corner of the video-viewer.
Resources

To retrieve a PDF copy of the presentation, click on the link below the slide player. From this page, click on the “Download” link to retrieve the file.
Presenters
Research Associate
School of Physiology, Pharmacology & Neuroscience
University of Bristol
School of Physiology, Pharmacology & Neuroscience
University of Bristol
Molecular Neuroplasticity
DZNE – German Center for Neurodegenerative Diseases
PhD Student
DZNE – German Center for Neurodegenerative Diseases
Group Leader
Institute of Experimental Medicine
Hungarian Academy of Sciences