Join Dana McTigue, PhD for a deep dive into her work studying spinal cord injury in rodents and its resulting pathological features in lower organs.
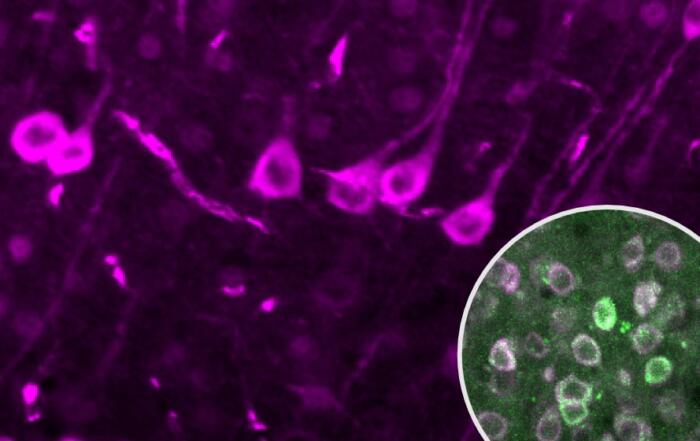
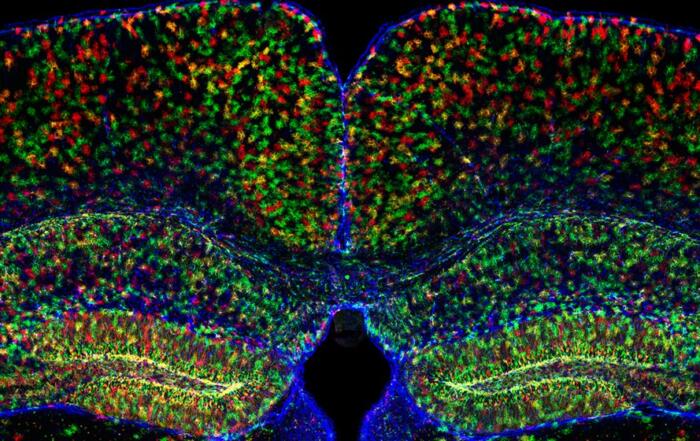
The spinal cord innervates the entire body at and below the neck. Thus, spinal cord injury (SCI) negatively impacts the spinal cord and all organs below the level of the injury. For more than 20 years, Dr. McTigue’s work has focused on glial repair after SCI, including oligodendrocyte progenitor cells (OPCs), which is shown to robustly divide and differentiate into oligodendrocytes that generate new myelin after SCI. Dr. McTigue and her lab’s recent work shows this surprisingly continues in mice for at least 6 months post-injury. Thus, the adult spinal cord remains highly dynamic for months after injury.
Prior mechanistic studies on regulators of OPC responses after SCI focused on iron, as it is essential for myelination. In studying iron chelation on OPC responses, they noted that the liver – the main iron regulatory organ in the body – was pathological after SCI. Following up this observation led the lab to discover that SCI causes rapid non-alcoholic steatohepatitis, an advanced form of fatty liver disease, as well as insulin resistance and hyperlipidemia, all of which are typically associated with metabolic syndrome and obesity. Current work is evaluating mechanisms to relieve metabolic pathology after SCI, as this is a prominent feature of clinical SCI in humans.
Key Topics Include:
- Understand that spinal cord injury negatively impacts the entire body beyond the lack of walking ability
- Understand what oligodendrocyte progenitors are and their role after spinal cord injury
- Understand that spinal cord injury causes metabolic syndrome in rodents and humans and that this contributes to the shortened lifespan in the SCI population
Presenters
Professor and Associate Dean
Neuroscience
Ohio State University