Unlocking Your Clinical Understanding of Post-Acute COVID Syndrome (PACS), Patient Recovery, and Risk of Subsequent Disease
Watch this webinar to learn about the metabolic changes during COVID-19 disease, how these can be accurately tracked and how NMR can improve the clinical understanding of Long-COVID.
Single-Domain Antibodies: A Promising New Tool for Diagnostic and Therapeutic Research
Dr. Bonnie Lun discusses applications and advantages of using single-domain antibodies for diagnostic and therapeutic research.
Informing Market Access Earlier in Clinical Development: Prospective RWE Research to Characterize Real World Patient Populations
Join Renee Willmon, MSc as she demonstrates Health Storylines as a disease-agnostic and customizable platform that connects healthcare stakeholders to engaged patients.
Solving SEC/GPC Limitations with Multi-Angle Light Scattering and Asymmetric-Flow Field-Flow Fractionation
Dr. Stepan Podzimek discusses the limitations of size-exclusion chromatography and how they can be solved using multi-angle light scattering and asymmetric-flow field-flow fractionation.
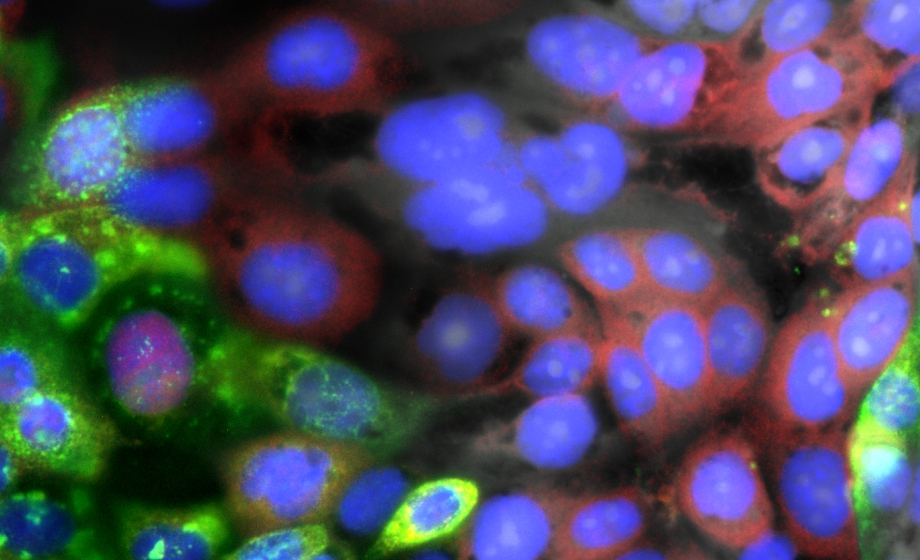
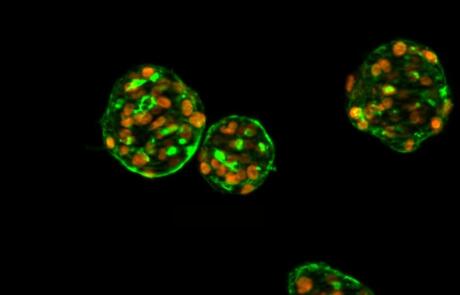
Visualizing the Intracellular Immune Response to SARS-CoV-2 with Fast, High-Content Imaging
Experts discuss how automated microscopy and image analysis can quantify critical markers of infection and describe the insight provided by microscopy in the context of SARS-CoV-2 infection and inflammation.
Using External Control Arm Studies in HTA Submissions: An Alternative, When RCTs are not Feasible
Matthias Hunger, Céline Taveau, and Ankit Pahwa present the challenges and opportunities when using external control arm studies in submissions to HTA agencies.
Overcoming Clinical Research Challenges: Analyzing EHR Data to Replicate Clinical Trials and Construct External Control Arms
Mac Bonafede, Stella Chang, and Dr. Rajesh Dash provide an overview on defeating common clinical trial barriers by combining EHR data with advanced analytics technologies.
The Value of a Multi-Source Real-World Data Strategy: How to Overcome Gaps and Limitations
Syapse experts share how to establish high quality, real-world datasets by using a multi-source strategy. A single source of data is often not enough to generate meaningful real-world evidence.
Value-Based Agreements – Are We Going to See More or Less of Them?
Mike Pace, MBA, Pierre Etienne, MD, and Clifton Chow give an insightful and practical webinar on one of the most important areas in pharmaceutical value and market access.
Can Literature Review Automation Improve Healthcare Cost-Effectiveness Analysis and Budget Impact Modeling Outcomes?
Michael Broder, President, PHAR and Jesse Ortendhal, Director of Health Economics, PHAR present on literature review automation in clinic.
Cutting Edge Conversations: Biomarkers – Challenges and Opportunities
The 5th and final episode in the Cutting Edge Conversation series for 2021 features Dr. Morad Nakhleh, Natalie Smith, and Dr. Bhavika Patel as they discuss industry challenges and innovations surrounding biomarkers in life science research and precision medicine
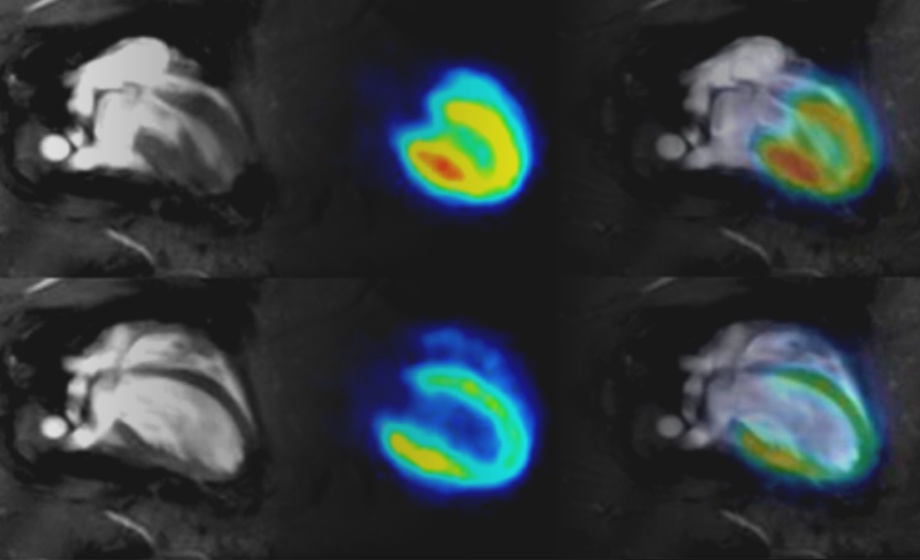
Cardiac PET/MR Imaging in Small Rodents
Experts provide a comprehensive overview of approaches to address cardiac and respiratory gating in PET and MRI in rodent models.
Cutting Edge Conversations: Innovative Approaches to Chemistry, Manufacturing and Controls
Episode 4 in the Cutting Edge Conversation series featured Dr. Mujeebur Rahuman, Dr. Holger von Moeller, and Dr. Fernando Huerta as they discuss about industry challenges and innovations surrounding Chemical, Manufacturing and Controls in the life sciences industry.
Translational Science Panel: Research Insights and Best Practice for Clinical Relevance
Join Aaron Phillips, Fiona McBryde, and Kevin O'Gallagher for a 1-hour Translational Science panel.
Cutting Edge Conversations: Value, Access and Evidence – Biotech Market Access
Sajjad Raza, Steven Fountain, and Sonya Snedecor present in Episode 2 of Scientist.com's Cutting Edge Conversations series with a presentation on the challenge research manufacturers face when they need to maximize access for patients with payers. The many considerations for a biotechnology company during various phases of drug development and how companies could use efficient sourcing strategies for market access.
Cutting Edge Conversations: Challenges in Sourcing Human Biosamples
Richard Kondo, McLean Collins, and Cathie G. Miller highlighted Episode 3 of the Cutting Edge Conversations series focused on innovations in the area of sourcing human biosamples for research. The event focused on sourcing human samples for research, including the value and necessity of working with human tissue during drug discovery, as well as some of the distinct challenges researchers face.
How Transformative Antibody Technology Can Improve Your Bioanalytical Assays
Dr. Royle discusses how the combination of antibody phage display and SpyTag technologies can generate critical antibody reagents with a difference.
Translational Applications of MiniPDX: An In Vivo Organoid Assay in New Drug R&D
Join Dr. Danyi Wen as she describes a novel drug efficacy test using Patient Derived Xenograft (PDX) samples to measure drug efficacy and identify therapeutic candidates in personalized oncology treatments.
Particulate Contamination in Medical Devices and Single-Use Systems
Join Mark Bumiller as he discusses the importance of subvisible particulate testing and provides an overview of solutions from Entegris to meet the qualification requirements.
Using Chemical Denaturants and Light Scattering to Determine Aggregation Propensity of Biopharmaceuticals
Dr. Luke Holloway reviews light scattering-based methods for protein characterization and explains their utility, when combined with partially denaturing buffers, in understanding aggregation propensity of monoclonal antibodies.
Plasma Proteomics: The Next Frontier of Biomarker Discovery in the Precision Medicine Era
Plasma proteomics is increasingly recognized as a fundamental approach to accelerate biomarker discovery and drug development, taking precision medicine to the next level.
SmartDriveNMR – The Next Generation of Automation
Bruker experts provide an overview of SmartDrive with a particular focus on the exciting new features. They offer practical examples and demonstrate the newest features and functions live!
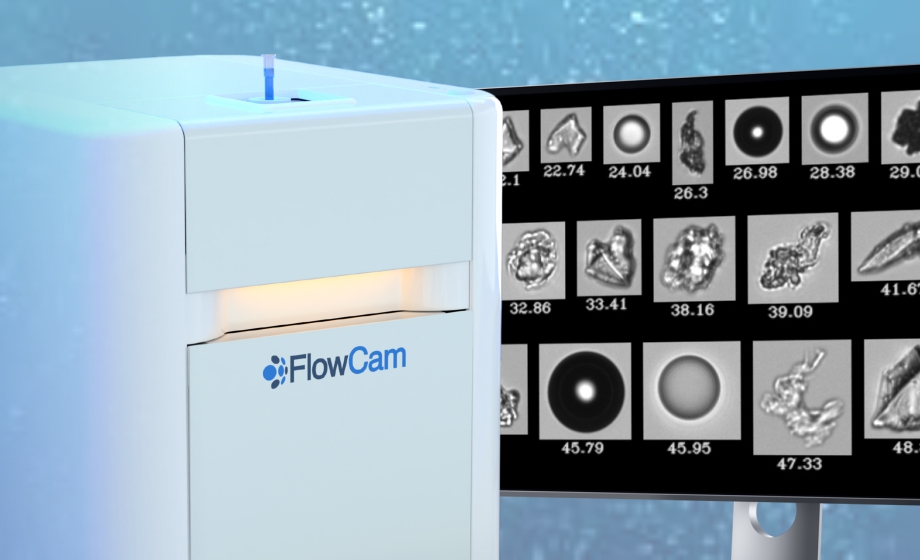
Detection of Protein Aggregation During Biopharmaceutical Development: The Role of Orthogonal Techniques
Dr. Danny Chou discusses the opportunities and challenges in orthogonal particle analysis, and how flow imaging microscopy and light obscuration can be used for formulation development and quality control.
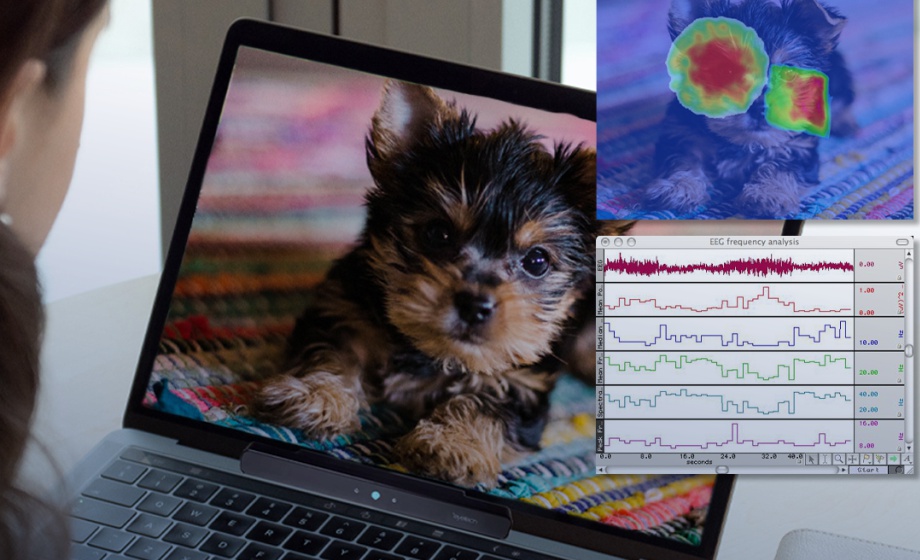
One Tool for Stimulus Presentation, Eye Tracking & Physiology Data
Experts discuss how to synchronize eye-tracking equipment with stimulus presentation, and physiological data.
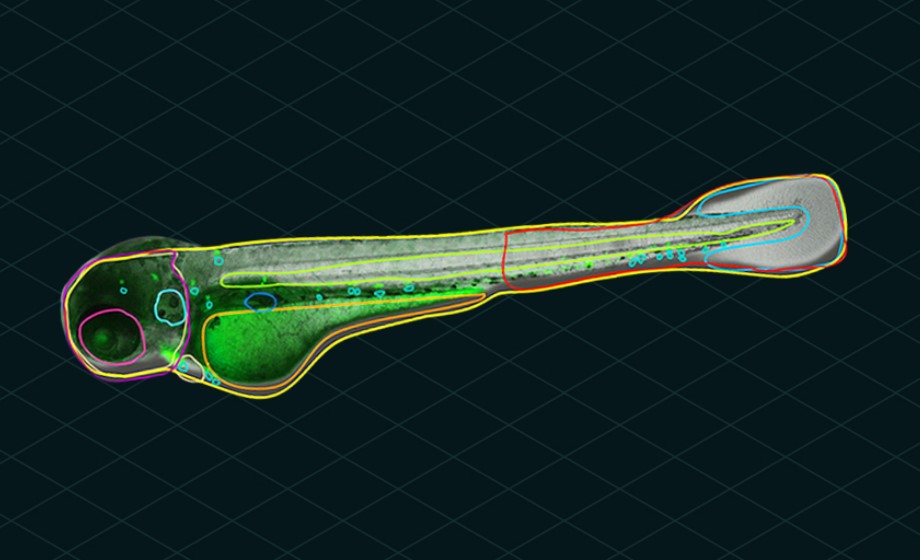
Casting a Wider Net in Zebrafish Screening with Automated Microscopy and Image Analysis
Join Dr. Alexandra Lubin and Dr. Jason Otterstrom as they discuss the use of deep learning-powered automated microscopy and image analysis for fast, in vivo zebrafish screening.